Accumulations of ammonia and nitrite are commonly observed in outdoor lined ponds, and elevated concentrations of these compounds during shrimp culture in a zero-water exchange system can cause shrimp mortality. Here are presented findings that enable estimations of total ammonia nitrogen (TAN) removal and oxygen net primary production (NPP) rates in an outdoor lined shrimp pond.
Outdoor aquaculture systems play a significant role in Thailand commercial shrimp cultivation because they entail low structural expenses and less operational complexity. Outdoor ponds with plastic liners have an advantage over traditional earthen ponds in terms of reducing soil pathogens, but the use of liners completely block the natural biological waste treatment processes that take place in soil-based ponds.

Biological degradation of unconsumed feed and solid or liquid animal excreta contributes to the buildup of toxic inorganic nitrogen waste, especially ammonia and nitrite. Accumulations of ammonia and nitrite are commonly observed in outdoor lined ponds, and elevated concentrations of these compounds during shrimp culture in a zero-water exchange system can cause shrimp mortality.
With the increasing popularity of outdoor high-density polyethylene (HDPE) lined ponds, especially in Thailand, proper management is critical for ensuring sustainable shrimp production. The study aimed to assess nitrogen removal, particularly total ammonia nitrogen (TAN), and microbic community dynamics in an outdoor HDPE-lined pond during a 76-day cultivation of Penaeus vannamei with zero-wastewater discharge.
Materials and methods
A semi-intensive culture of Pacific whiteleg shrimp, P. vannamei at a stocking density of 40 shrimp/m2 was operated in an outdoor pond that was lined with 0.7 mm HDPE. The location was a demonstration shrimp farm owned and operated by the Chaipat tana Foundation in Chachoengsao, Thailand.
Chlorination was used for water disinfection prior to stocking. Operation periods and feeding rates could be used to divide the 76-day shrimp cultivation into three phases. The experiment was continuously operated without water exchange under the following conditions: dissolved oxygen (DO) of >5 mg/L and alkalinity of >100 mg/L as CaCO3 by sodium bicarbonate supplement.
Results
In Phase I (days 1–30) of shrimp cultivation, the feeding rate gradually increased from 0.5 to 9.5 g/m2 d and accounted for the estimated nitrogen feed loading of 0 to 0.5 mg-N/L d. During this time, low inorganic nitrogen levels were observed as 0.0 ± 0.0 mg-N/L for both TAN and nitrite and as 1.2 ± 0.3 mg-N/L for nitrate.
In Phase II (days 31–50) feeding rate was increased to 8.2 ± 1.2 g/m2 d (equal to 0.4 ± 0.1 mg-N/L d) and in Phase III (days 51–76), it was increased to 11.3 ± 2.1 g/m2 d (equal to 0.6 ± 0.1 mgN/L d).
The nitrogen waste loading was estimated to be 48.0 kg-N/t-production. TAN levels increased for a week in Phase II (0.1–0.4 mg-N/L; days 38– 44) and then decreased to <0.1 mgN/L. Similarly, in Phase III, TAN levels increased from 0.5 ± 0.0 mg-N/L (day 63) to the maximum allowable level of 1.0 ± 0.0 mg-N/L (day 70).
Nitrite was not detected throughout the culture. The average DO concentration was 5.6 ± 1.2 mg/L, reaching 9.6 mg/L during the daytime. The temperature ranged from 25.6 to 30.8ºC, with an average of 28.9 ± 1.1ºC. Chlorophyll levels increased as the cultivation proceeded.
Increases in TSS levels were detected throughout cultivation. During the first three weeks, TSS was relatively low (61 ± 17 mg/L), but the TSS level almost tripled to 169 ± 11 mg/L on day 27 and stayed relatively high at 124–186 mg/L in Phases II and III.
The TAN removal rate indices increased as the cultivation age increased in both control (pond water) and treatment (pond water plus plastic sheet) chambers (Figure 1).

Significantly increased TAN removal rates were observed in Phase III in which rates by day 55 had rapidly increased to 0.22 ± 0.2 g-N/m3 h from day 48 (p ≤ 0.05, ANOVA). Thereafter, they remained stable to day 62 (0.21–0.24 g-N/m3 h). By day 70, the rates had significantly reduced from day 62 and there was a significant difference between the TAN removal rates of the control and treatment groups.
A significant increase in oxygen NPP (1.16 ± 0.16 g-O2/m3 h) had occurred by day 48 (p ≤ 0.05, ANOVA). Sediment samples contained 4.06 ± 0.16% carbon, 6.89 ± 0.34% hydrogen, 1.79 ± 0.07% nitrogen and 1.63 ± 0.01% sulphur. The total nitrogen loading in the shrimp cultivation crop was 38.7 g-N/m2 (Table 1).
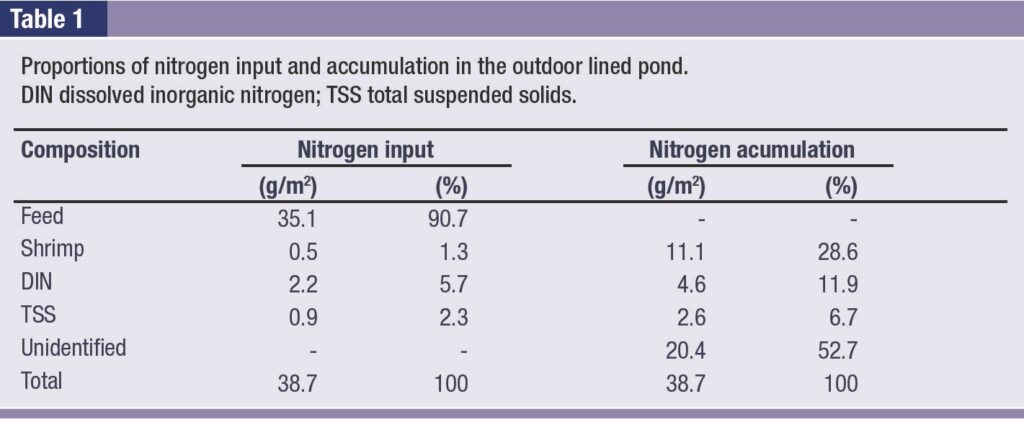
Biofilms of microbial communities were observed on almost all the plastic liner samples examined. These could have been photosynthetic or non-photosynthetic bacteria or algae. Some biofilms included filamentous organisms of 3.47 ± 0.30 μm diameter.
These filamentous organisms could also be photosynthetic or nonphotosynthetic bacteria or algae. Principal component analysis (PCA) revealed a separation among the bacterial profiles of weekly collected water samples on the first two components.
The bacterial profiles of pond water shifted from days 1 through 75. Phase I (days 20 and 28) showed water bacterial profile separation from Phases II and III on the first axis (34% of the variances), and Phase II (days 33, 34 and 48) tended to separate from Phase III (days 62 and 75) on the second axis (20% of the variances).
Interestingly, the samples of Phase III (days 62 and 75) was clustered closer together than the samples of any other adjacent week pairs.
Figure 2 shows a significant positive correlation between increases in TAN concentration and the relative abundances of Chlorophyta, Cryptophyta, and Euglenozoa, but a significant negative correlation with the abundance of Bacillariophyta (p ≤ 0.01, Pearson’s correlation).
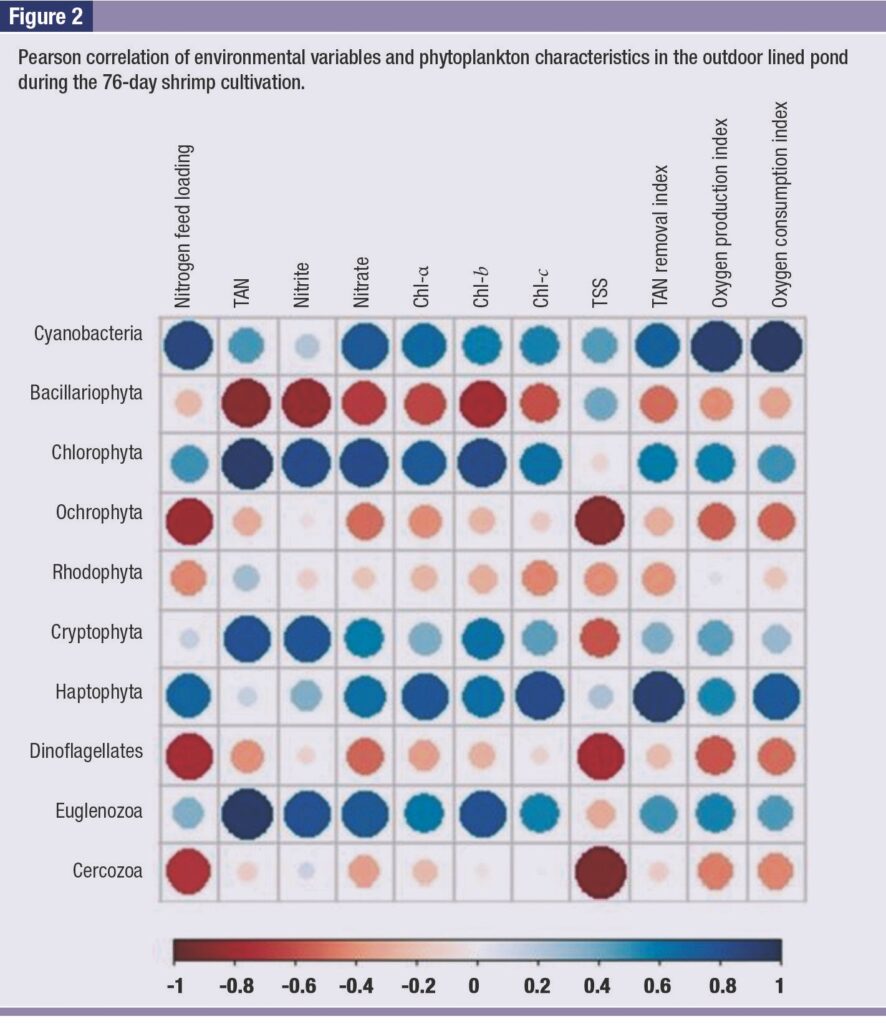
Furthermore, increases in nitrate were found to have a significant positive correlation with the relative abundances of Cyanobacteria, Chlorophyta, and Euglenozoa (p ≤ 0.05, Pearson’s correlation).
Discussion
The biological processes of phytoplankton and other microorganisms actively maintained low nitrogen waste levels throughout the 76-day shrimp cultivation crop. TAN was largely eliminated by assimilation processes because there was no detectable nitrite that would be expected to result from TAN oxidation.
A dominant assimilation process has typically been reported in outdoor aquaculture systems via the photoautotrophic uptake of TAN.
“This is because phytoplankton growth is favorably promoted by available nutrients, especially inorganic nitrogen and inorganic carbon sources (Gerardi and Lytle, 2015; Lemonnier et al., 2017; Saraswathy et al., 2012).”
Additionally, the observed small reduction in nitrate levels in Phase I could have resulted from both photoautotrophic uptake and denitrification via heterotrophic bacteria (Burford et al., 2003; Ebeling et al., 2006; Hargreaves, 2006).
The occurrence of anoxic denitrification activities, in the sludge layer on the HDPE liner surface, was indicated by the negative oxidation reduction potential (ORP) values of less than –200 mV, which signifies a mildly anoxic environment is conductive to denitrification.
These values were close to the ORP values observed in the bottom substrate denitrification treatment tank (Pungrasmi et al., 2013). The accumulation of such solid deposits at the center of the pond bottom is known to result from paddlewheel operation and leads to oxygen depletion inside the cumulated sediments.

During Phases II (days 31–50) and III (days 51–76) of cultivation, the outdoor lined shrimp pond transitioned to a mixotrophic system. Photoautotrophic uptake continued to govern inorganic nitrogen concentrations, as evidenced by the stable TAN uptake rate indices during the daytime.
Moreover, it is notable that heterotrophic or mixotrophic microorganisms can contribute to the uptake of ammonia, even in the absence of light (Burford et al., 2003). Previous studies conducted in outdoor aquaculture ponds have shown that phytoplankton contributes more significantly to TAN removal than bacteria.
“Intermittent increases or decreases in alkalinity were detected in Phases I and II, but the obvious decline in Phase III might have been associated with the rapid increase in measurable chlorophyll levels.”
The dominant activities of phytoplankton were supported by continuously increased abundance of Cyanobacteria, which were positively correlated with chlorophyll levels.
Furthermore, Cyanobacteria was also found to have a positive correlation with TAN availability. This finding is in accordance with a previous study reporting that their growth is typically linked to ammonia levels from shrimp nitrogen waste excretion and the biological degradation of organic nitrogen (Domingues et al., 2011).
Conclusions
The maturation of the pond ecosystem, in the outdoor HDPE-lined shrimp pond with no water discharge, was observed to progress from the photoautotrophic (days 1–30) to the mixotrophic phases (days 31–76). The consistency of the estimated TAN uptake rate index, which was calculated based on photosynthesis, confirmed the outstanding activity of photoautotrophic uptake in controlling inorganic nitrogen waste.
Subsequently, the difference between the photoautotrophic uptake and the overall TAN removal rate indices, in the pond water suggested the involvement of bacteria in the nitrogen cycle.
“The overall nitrogen waste carrying capacity of the lined pond was largely supported by the activities of suspended phytoplankton in the water, especially cyanobacteria and other phytoplankton, and was much more influential than the activities of attached organisms on the plastic liner.”
Based on chloroplast analysis, Bacillariophyta was the predominant chloroplast in the shrimp pond, while the abundance of Chlorophyta enhanced with prolonged operation and was positively correlated with the TAN level.
In addition to photoautotrophic uptake, the ORP confirmed the role of nitrate elimination through anoxic denitrification in aggregated sediments at the bottom of the pond, despite the absence of soil.
This article is sponsored by: REEF INDUSTRIES INC.

This is a summarized version developed by the editorial team of Aquaculture Magazine based on the review article titled “TOTAL AMMONIA NITROGEN REMOVAL AND MICROBIAL COMMUNITY DYNAMICS IN AN OUTDOOR HDPE-LINED SHRIMP POND WITH NO WATER DISCHARGE” developed by: Satanwat, P. and Tapaneeyaworawong, P. – BIOTEC, Chulalongkorn University; Wechprasit, P. – BIOTECM, Mahidol University; Boonprasertsakul, T. – BIOTEC, Chulalongkorn University; Pungrasmi, W. – Chulalongkorn University; Sritunyalucksana, K. and Prachumwat, A. – BIOTEC, Mahidol University; Powtongsook, S. – BIOTEC, Chulalongkorn University, Marel-website-news.
The original article, including tables and figures, was published on JUNE 2023, through AQUACULTURE.
The full version can be accessed online through this link: https://doi.org/10.1016/j.aquaculture.2023.739898.









