There is a growing recognition of the challenges associated with ensuring good nutrition for all without compromising the environment. This is particularly true for aquaculture, given the reliance on marine extraction for key feed ingredients, yet at the same time it delivers key nutrients such as omega-3 long chain polyunsaturated fatty acids. This review will consider progress in transitioning away from oceanic-derived fish oils as feed ingredients, focusing on the emerging transgenic plant sources of these fatty acids.
Introduction
The health-benefits associated with a diet containing oily fish rich in omega-3 long chain polyunsaturated fatty acids (omega-3 LC-PUFAs) are now well-established, linking regular consumption of these fatty acids with reduced risk of cardiovascular disease and myocardial infarction.
However, many factors have conspired to make this apparently easy recommendation less easy to fulfil, including the relentless growth in the global population which puts increasing demands on our planet’s finite resources, pollution of the marine environments and advancing climate change.
“Perhaps counterintuitively, the growth in aquaculture as the dominant farming system to produce fish for human consumption, has altered the dynamics of omega-3 supply and demand, putting additional strain on finite global fish stocks.”
As discussed below, efforts to make aquaculture more sustainable have lowered the nutritional value of the final product. In this sense, the inclusion of terrestrial plant oils, the main alternative to fish oil in Europe, in aquafeeds has led to a marked reduction in the levels of omega-3 LC-PUFAs accompanied by increased levels of short chain fatty acids characteristic of terrestrial systems.
Long established dietary patterns have radically shifted in the last few decades, to the point that metabolic diseases resulting from the overconsumption of calorierich but low nutrient diets outweighs (no pun intended) the number of people suffering from poor nutrition due to insufficient food.
“Simultaneous to that, a growing awareness that global patterns of food consumption are exceeding planetary boundaries has also led for calls in a radical shift in how we feed ourselves, most notably with a strong decrease in the consumption of (terrestrial) animal protein and concomitant increase in dietary plant protein.”
Interesting, elevated consumption of fish is also recommended (because of the nutritional benefits of omega-3s etc.) but there is an inherent tension associated with such advice, not least of all with respect to sustainability of global fish stocks.

Plant biotechnology has always aspired to deliver benefits to society, even if this has sometimes not been obvious. The first wave of GM crops developed in the 1990s was focused on delivering so-called input traits, which help farmers (via herbicide tolerance and/or herbivore resistance) but provide no direct benefit to the consumer or nutritional enhancement (although they do deliver a reduced reliance on chemical inputs).
“A second wave of output traits for improved nutrient content is only now starting to appear, 25 years behind the entry-level features are the many and varied reasons for this have been studied recently by us and others.”
At the forefront of these new GM crops are plants which have been engineered to accumulated omega-3 LC-PUFAs in their seed oils, providing a novel terrestrial source of this important fatty acids independent of marine stocks − such plant-based sources of “omega-3 fish oils” can be used for both direct human nutrition or as animal feed ingredients (in either aquaculture or terrestrial animals), delivering benefit by these different routes.
The omega-3 LC-PUFAs, eicosapentaenoic acid (EPA; 20:5∆5,8,11,14,17) and docosahexaenoic acid (DHA; 22:6∆4,7,10,13,16,19) are found in fish oils and are almost exclusively restricted to aquatic foodwebs. The predominant biosynthesizers of EPA and DHA in the marine ecosystems are microalgae and other phytoplankton, with the fatty acids accumulating in the multiple trophic levels above these founders.
“Marine and salmonid fish species have little or no endogenous capacity to synthesise these fatty acids, relying instead on dietary intake. No angiosperm plant species have the capacity to synthesise EPA and DHA, although C18 precursor polyunsaturated fatty acids are ubiquitous in the Plant Kingdom.”
Since the 1990s, significant efforts have been made towards engineering plants to accumulate EPA and/or DHA as an alternative to marine extraction. Representing one of the very few projects to progress first from a discovery phase into application, the production of omega-3 fish oils in transgenic plants represents a significant achievement for plant biotechnology.
This short review will concentrate on recent examples of scale-up and translation, including demonstration of efficacy in aquafeed trials. Aspect of the molecular biology and genetic toolkits that have been used to generate these transgenic plants have been recently considered elsewhere and are therefore only discussed briefly.
Transgenic plants as a platform for the synthesis of EPA + DHA
Despite being a deceptively straightforward objective, the efficient synthesis of the non-native fatty acids EPA and DHA proved challenging to achieve, especially when accumulation was restricted to storage triacylglycer ols via seed-specific promoters.
This was due in part to the relative complexity of the biosynthetic pathway, requiring the co-ordinated expression of a minimum of five heterologous genes, representing some of the most complex metabolic engineering at tempted in transgenic plants.
How ever, by adopting an iterative approach to defining the optimal combination of genes and regulatory elements, significant progress has been achieved in two host species, Camelina and Canola.

Interesting, it has also become apparent that endogenous metabolism plays a key role in enabling the effective reconstitution of the omega-3 LC-PUFA biosynthetic pathway and this inherent capability varies between species. For example, the successful synthesis of EPA + DHA in Arabi dopsis or Camelina via the so-called alternative pathway was not recapitulated in peanut.
“Even in the case of effective oil seed chasses such as Canola and Camelina, there are significant differences between the species e for example, a very similar suite of genes and regulatory elements was used to direct the synthesis of EPA + DHA, resulting in ⁓20%EPA + DHA in Camelina but only 8% EPA and 1% DHA in Canola.”
This may indicate additional levels of control, which could include transgene integration site, post-transcriptional regulation, enzyme turnover and substrate channeling.
Independently of these open questions, it is clear that Brassicacea oilseeds are excellent hosts for the transgenic accumulation of omega-3 LC-PUFAs (summarized in Table 1) and the oils derived from these crops have been used in multiple aquafeed trials.
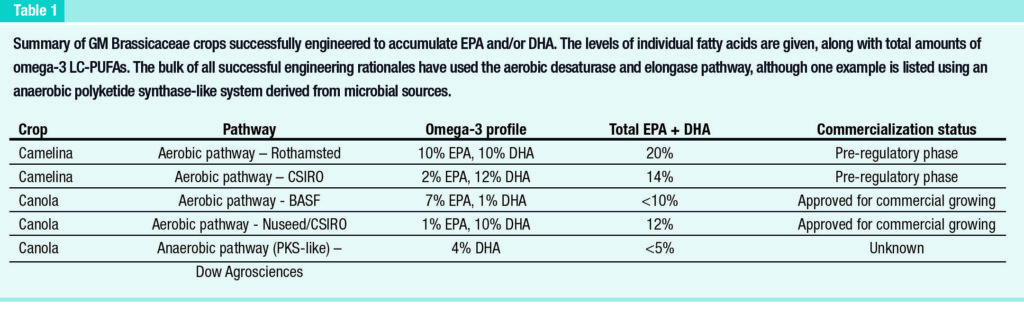
Importantly, the realworld performance of these crops has been demonstrated by multi-location field trials across different continents. Recently, gene-editing has been used in conjunction with transgenesis to enhance the accumulation of EPA + DHA in Camelina via the inactivation of a competing endogenous pathway.
Novel oils in aquafeed – validation in fish feeding trials
One key aspect of achieving the successful translation of the biotech studies described above is to validate these novel oils as effective substitutes for fish oils in the diets used in aquaculture (represented in Figure 1).

To date, the majority of research in fish using GM-derived oils rich in n-3 LC-PUFA has predominantly focused on Atlantic salmon (Salmo salar).
This is largely due to the fact that salmonids are the primary consumer of extracted fish oils given the energy dense feeds required for farming, as well as its high-end value. Therefore, it is of paramount importance that the potential effects of using such substitutes are investigated.
Consequently, oils derived from both omega-3 rich Camelina and Canola (Event NS-B50027-4) have been successfully trialed in several life stages.
Freshwater fingerlings fed either 4 or 8% high n-3 GM Canola oil (9.3% omega LC-PUFA) exhibited similar growth and survival to those fed a diet containing fish oil as the main dietary lipid source when cultivated at either 12 or 16 oC.
In seawater, no difference in growth were found in smolts fed a Camelina oil rich in either EPA or EPA + DHA (23.9% and 16.7% omega-3 LC-PUFA, respectively) compared to those fed a fish oil rich diet.
“Moreover, all trials showed that the fish fed the GM-derived oils had enhanced levels of n-3 LC-PUFA in their flesh compared to either a commercial diet control or a feed containing wildtype Camelina, while also decreasing the omega-6:omega-3 ratio.”
For example, salmon fed a diet containing high omega-3 LC-PUFA Camelina oil accumulated almost double the amount of these health beneficial fatty acids as salmon fed a fish oil diet. Evidently, these enhanced levels can contribute to bridging the gap between demand and supply and help meet the 500 mg/ day intake requirement for omega-3 LC-PUFA in order to attain optimal cardiac health.
Enhanced flesh levels of EPA + DHA has been a common finding in other studies where teleost fish were fed omega-3 LC- PUFA vegetable oils such as other fatty species including rainbow trout (Oncorhynchus mykiss), but also gilthead sea bream (Sparus aurata) and sea bass (Dicentrarchus labrax).
“In addition to the benefits that the use of these oils can have for consumers, the use of vegetable oils rich in omega-3 LC-PUFA can also have added benefits for the fish such as reduced liver fat infiltration and hepatocyte area relative to fish fed feeds containing wildtype Camelina.”
Perhaps, most importantly to note is that none of the trials performed to date have reported any adverse effect in fish fed these novel oils thus demonstrating the potential for the exploitation of this key raw material. Studies so far have focused on the use of the oil, whereas the seed cake has yet to be tested as an alternative protein source.
Additional processing of the seeds such as dehulling or solvent extraction might be required to concentrate the protein fraction which in turn will increase the price as well as the environmental footprint of the product.
Next steps and new approaches
The validation of these novel oils as safe and effective substitutes for oceanic-derived fish oils was a key step in derisking this technology and helping it advance to market. In addition, increased awareness of issues associated with sustainability and environmental footprint have added impetus to commercialization.
“Exciting, two Canola events developed by BASF/Cargill and Nuseed/CSIRO have been deregulated by USDA and now can be cultivated at scale.”
These events represent the most complex metabolic engineering in plants to have been approved for commercial cultivation to date, although these developments represent the culmination of 25 years of fundamental research followed by a concerted focus on development.
Simultaneously, oil from the lead Camelina event has been used extensively in human studies, confirming the utility of this oil in both indirect (i.e., animal) and direct human nutrition.

There are also efforts to combine a number of different traits to produce a multifunctional crop tailored to the needs of e.g. aquaculture, combining the EPA + DHA trait with other high value components of aquafeed diets such as the antioxidant and pigment astaxanthin, which can be made in a number of different transgenic hosts and has been independently validated as a novel ingredient in fish feeding trials.
Similarly, the amino acid composition and seed storage proteins can be enhanced to make the resulting protein composition more suitable for fish nutrition (e.g. by the inclusion of the cysteine derivative, taurine) as has been achieved in transgenic soybean.
“Equally importantly, it is possible to use gene editing to remove compounds such as glucosinolates which can act as feeding deterrents for some fish species.”
On this basis, it is possible to envisage a tailor-made cultivation ideotype specifically designed for the nutritional needs of both the fish and the human consumer whilst also delivering to the requirements of operating within planetary boundaries and leaving a lighter environmental footprint on our planet.
Conclusions
Recent analysis confirms the significant variance between demand for omega-3 long chain PUFAs and the available supplies from our oceans, currently estimated at providing only ⁓30% of the recommended levels of EPA and DHA for the global population.
This highlights the pressing need for alternative supplies of these important fatty acids, not least of all given their health-protective role against cardiovascular disease and other metabolic pathologies which now blight the lives of many millions of people.
“In addition to the sustainable synthesis of EPA & DHA via transgenic plants, a number of microbial (predominantly algae) systems are being established and also following the same path of validating these novel oils via aquafeed trials.”
One advantage of using algal-derived omega-3 PUFAs is that these strains are usually not GMOs, making their regulatory approval and consumer acceptance relatively straightforward. However, the largescale culture of algal strains remains a challenge, in terms of economics and infrastructure.
Moreover, almost all commercial algal platforms rely on heterotrophic growth (i.e., cultured using an exogenous carbon source which itself is invariably of vegetable origin, such as sugar cane) and this is not always fully reflected in sustainability declarations and it would be interesting to carry out a life cycle analysis of the different production systems to allow the consumer to make an informed choice with regard to environmental footprint.
“Other sources of LC-PUFA such as krill oil are now mainstream ingredients in aquafeed, although their inclusion is normally limited mainly due to its high price.”
Another approach to boost the levels of these health-beneficial fatty acids in fish without impacting on fisheries is the use of finishing diets high in LC-PUFA. Although the novel plant platforms described above are GM with the associated regulatory burdens, they have the advantage of utilizing the pre-existing infrastructure and know-how which underpin modern agriculture, allowing rapid scaling and incorporation into production cycles.
Expanding our use of plant-derived feed and foodstuffs will be essential for operating within planetary boundaries and collectively we need to embrace a different approach to ensuring optimal nutrition for all e plant biotechnology can play a key role in enabling this.
As such, it is important to consider all ethical aspects of using GM-derived ingredients, including the impact of not adopting these innovations.
This is a summarized version developed by the editorial team of Aquaculture Magazine based on the review article titled “ENGINEERING PLANT-BASED FEEDSTOCKS FOR SUSTAINABLE AQUACULTURE” developed by: NAPIER, J.-Rothamsted Research; BETANCOR, M.-University of Stirling.
The original article was published, including tables and figures, on FEBRUARY, 2023, through PLANT BIOTECHNOLOGY.
The full version can be accessed online through this link: https://doi.org/10.1016/j.pbi.2022.102323.









