Shrimp has been recognized as the second most traded aquaculture products for decades. However, shrimp production in major producing countries has declined, mostly due to disease problem. Here we present a study aimed to determine the effect of dietary mannan oligosaccharides delivered through Artemia sp on the survival, growth, immunity, digestive enzymes and resistance against Vibrio harveyi of Pacific white shrimp larvae.
Shrimp has been recognized as the second most traded aquaculture products for decades. During the recent years, however, shrimp production in major producing countries has declined, mostly due to disease problem (FAO, 2016). Stentiford et al. (2017) recently reported that infectious diseases have caused shrimp industry to suffer devastating economics and social impacts with total losses exceeding 40% of global capacity.
Amongst the efforts have been done to alleviate this problem, avoidance of disease outbreaks by management of pond and animal microbiomes remains to be the most feasible approach to mitigate losses rather than attempting to eliminate the presence of pathogens.
“In this regard, the strategies to address disease problems in shrimp production have been directed toward improving the culture environment and enhancing the cultured animal’s strength and defense against disease infection.”
The later include improvement of the animal’s health status by supplying sufficient nutrients to support good growth and robustness, applying vaccination and immunostimulation to improve immunity and conditioning the animal’s microbiome by probiotics and prebiotics application.
Mannan oligosaccharides (MOS) are one of the prebiotics groups that have been gaining attention for the application in aquaculture, which are glucomannoprotein complexes derived from the cell wall of yeast, Saccharomyces cereviceae.
“Some beneficial effects of MOS have been recorded so far include enhancing health and growth performance improving gut morphology and microbiota, increasing immunity against disease; and robustness against stress.”
Despite the growing evidence of prebiotic beneficial effects on the growth, survival and immune response of various aquaculture species, there are only few studies have been done to explore the effects of prebiotic encapsulation in live food on the larviculture of aquaculture species.
This study used Artemia to facilitate the delivery of MOS to the larvae, as it is the most common live food used in shrimp hatchery, which has a compatible size and nutritionally adequate for the larvae.
Materials and Methods
Experimental Design and Shrimp Rearing
This study aimed to determine the effect of dietary mannan oligosaccharides (MOS) delivered through Artemia sp at different concentrations (i.e. 0, 3, 6 and 12 mg L-1) on the survival, growth, immunity, digestive enzymes and resistance against Vibrio harveyi of Pacific white shrimp larvae in four treatment groups (in triplicates), i.e. control, MOS3, MOS6 and MOS12.
Larvae were reared in tank with a dimension of 25 cm × 20 cm × 30 cm containing 4 L of disinfected seawater at a stocking density of 30 larvae L-1. The larvae were maintained until they reached post larval stage for 15 days and were subsequently challenged with V. harveyi (107 CFU mL-1) for 5 days.
Artemia Enrichment
Artemia enrichment was performed following the procedures described by Daniels et al. (2010) with some modifications. Artemia naupli was obtained by hatching 2 g of Artemia cyst (Supreme Plus, US) in 1 L of seawater. Enrichment was performed by adding MOS (BIOMOS, Altech Inc., KY USA) into newly hatched Artemia nauplii culture medium (seawater at a salinity of 30 gL-1) at a concentration depending on the treatment.
“Artemia nauplii were maintained in prebiotic suspension at a density of 100 individual mL-1 for 4 h. Similar procedure was applied to Artemia nauplii provided for the shrimp in control treatment, except there was no prebiotic added to the Artemia culture medium.”
The nauplii were subsequently collected using a plankton net, rinsed with fresh seawater and directly transferred to the shrimp larvae culture tank or kept in a refrigerator (4°C) for later feeding time that day. Shrimp survival, growth, digestive enzymes activities, bacterial count, immune parameters were determined at the final day of experimentation.
Shrimp survival was the proportion of survived shrimp at the end of the experiment over the initial number of shrimp at the beginning of the experiment.
Challenge test
Upon the completion of the feeding experiment, challenge test was performed using pathogenic Vibrio harveyi MR5339 isolated from shrimp infected with luminescent vibriosis (a collection of the Fish Health Laboratory of the Department of Aquaculture, Bogor Agricultural University).
“Challenge test was carried out on 10 shrimp from each replicate tank placed 15 units of glass container previously filled with 1 L of disinfected seawater. The bacterial suspension was added to each container at an initial density of 107 CFU mL-1.”
On the following day, V. harveyi suspension was occasionally added to replace loss due to water replacements, which were done on day 1 and 3 of the challenge test period. Feeding during challenge test was done using non-enriched Artemia nauplii, which was offered five times a day.
Negative control using the shrimp from the control treatment was applied in this test with similar treatment, except no V. harveyi suspension was added to the container. Challenge test was performed for 5 days.
Immune Parameters
Immune parameters were measured after feeding experiment (prior to challenge test) and after the completion of challenge test. Body fluid collection was conducted following the procedures previously described in Tampangallo, Pakidi, and Rantetondok (2012) with some modifications.
Briefly, about 0.3 g of larvae (five to six post larvae) were collected from each replicate tank, placed in a pestle and added with 900 µL of precooled anticoagulant solution. Thereafter, each larva was slightly compressed to let the haemolymph flowing out the shrimp’s body and got mixed with anticoagulant solution.
Results
Larvae Growth Performance
The highest shrimp survival was observed in MOS12 treatment, which was significantly different from other treatment groups (p < 0.05). MOS administrations on shrimp larvae resulted in higher growth as indicated by the significantly higher SGR and shrimp final length (Table 1, p < 0.05). The highest growth was observed in shrimp in MOS12 treatment.

Digestive Enzyme Activities and RNA/ DNA Ratio in Shrimp Larvae
The digestive enzyme activities in shrimp larvae fed with MOS were higher than those in the control group (Table 2). Furthermore, the increase of digestive enzyme activity was positively correlated with the increase of MOS concentrations with correlation coefficients for protease, lipase and amylase were 0.96, 0.95 and 0.91, respectively (p < 0.05).

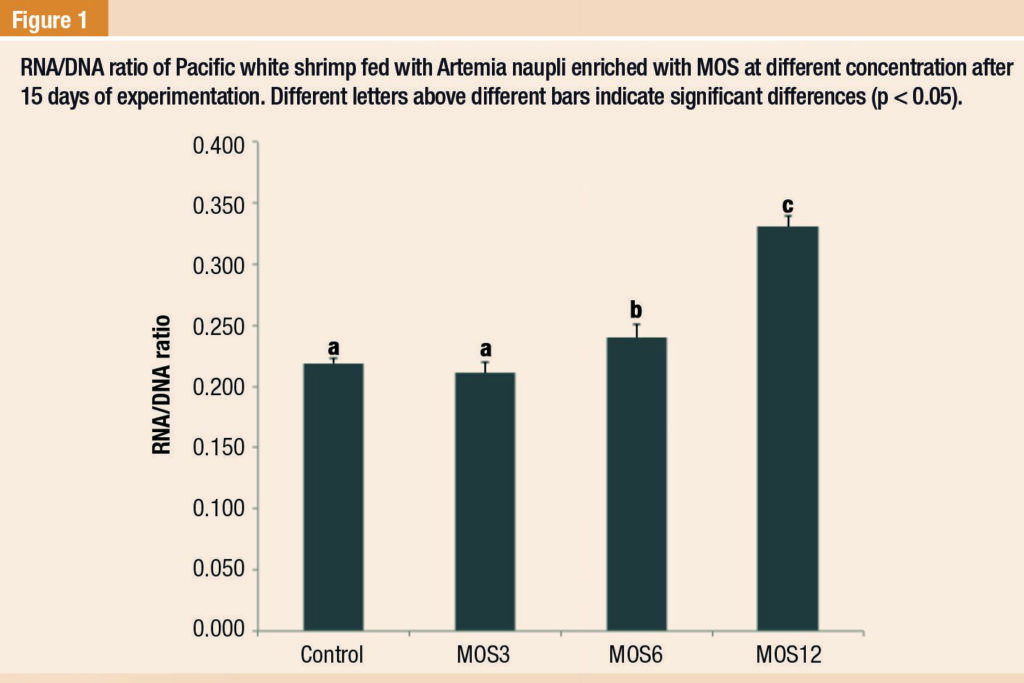
The ratio of RNA/DNA in shrimp with treatment MOS6 and MOS12 were significantly higher than those with the control and MOS3 treatment (p < 0.05) (Figure 1).
Immune Parameters and Post Challenged Survival
After feeding experiment, the immune parameters in treatments with MOS were evidently improved, with the levels increased with the increase of MOS concentrations (Table 3). After challenge test, decreases in all immune parameters were observed compared to those before challenge test.
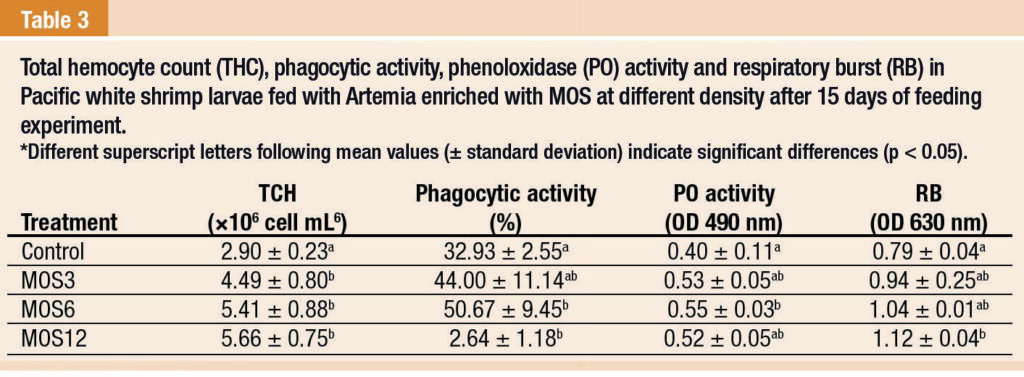
However, the shrimp immune parameters before and after challenge test showed similar trends, where shrimp fed with MOS-encapsulated Artemia nauplii showed higher levels of immune responses (Table 4).

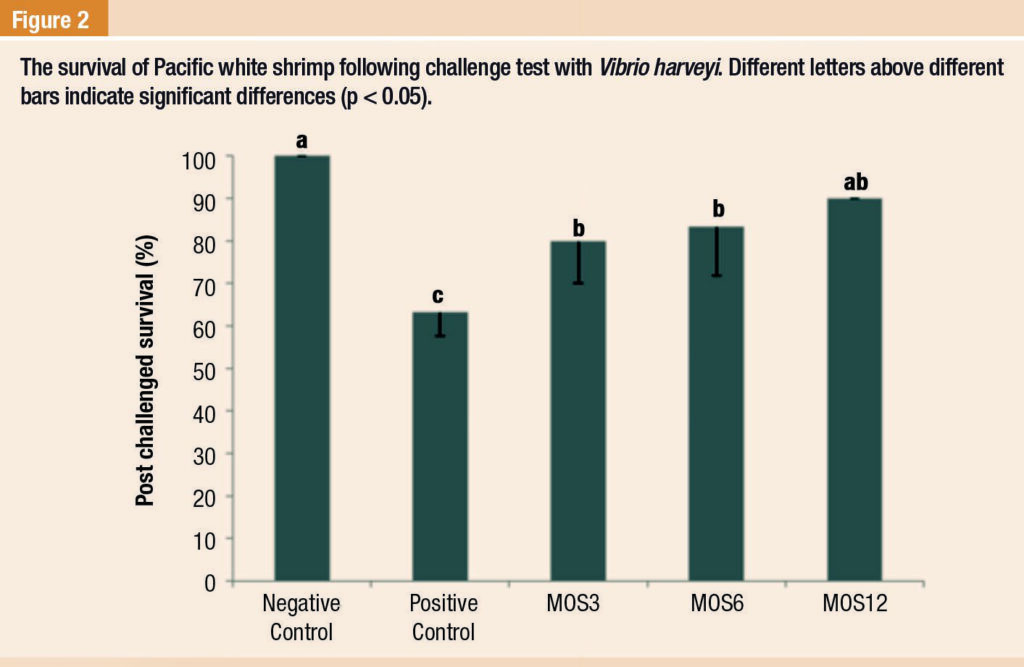
This was confirmed by the higher number of shrimp survivors in MOS treatments than that of the positive control following challenge test against V. harveyi (Figure 2).
Discussion
To date, most of MOS or other prebiotics studies were performed in juvenile or adult (grow out) stage and only limited information is available on their application in larvae production of aquaculture species. Thereby more research is needed to fully comprehend the efficacy of dietary prebiotics in crustacean larvae production.
The application of MOS on crustacean larvae production has been reported so far was done by Daniels et al. (2010; 2013) who demonstrated the positive effects of MOS-enriched Artemia on the growth and immunity of European lobster postlarvae.
“The present study demonstrated that MOS encapsulation using Artemia nauplii positively affected the growth performance of shrimp larvae.”
Although the differences between MOS3 and MOS6 treatments were not significant, it was generally shown that the increase of prebiotic concentrations was concomitant with the increase of shrimp specific growth rate and final total length.
Furthermore, the differences in growth potential of the shrimp post larvae amongst the treatments were also demonstrated by the levels of RNA/DNA ratio as an indicator of protein synthesis (Li et al., 2013). The positive effects of MOS in improving growth performance were consistent with previous study by Zhang et al. (2012) on Pacific white shrimp juveniles.
The underlying modes of actions of dietary MOS on the animal’s growth have been reported to this point mostly relates to:
1) the improvement of gut morphology and functionality, i.e. increase of microvilli length and density, higher number of goblet cells, higher production of intestinal mucus, which may facilitate higher capacity for nutrient digestions and absorptions in the animal’s intestine (Sang et al., 2011a; Zhang et al., 2014),
2) the facilitation of beneficial microbiota in the animal’s gastrointestinal tract (GIT) that may contribute to the excretion of digestive enzymes and to the alteration in host’s nutrients metabolism;
3) the improvement of feed intake by contributing in satiation and appetite signals and regulations (Torrecillas et al., 2014).
Indeed, this study demonstrated that dietary supplementation of MOS could improve the activity of main digestive enzymes, i.e. protease, lipase and amylase, which may contribute to the increase capacity of nutrient digestion and ultimately resulted in better growth of the shrimp postlarvae.
Previous studies showed that the effect of MOS on digestive enzymes might be different between species or between developmental stages. Sang et al. (2011a) showed that MOS could improve the digestive enzyme activity in hepatopancreas and midgut of yabbies (Cherax destructor), thus increasing the bioavailability of nutrients in the diet.
“However, further study is needed to elucidate the role of prebiotics on the digestive enzyme activity.”
The enhanced shrimp survival in MOS treatments might be explained by the enhanced shrimp robustness against stress and immunity against possible disease infection. Indeed, the results showed that following 15 days of dietary MOS treatments, the shrimp total haemocyte count, phagocytic and PO activities and respiratory burst were significantly enhanced.
These results confirmed previous study showing that dietary administration of MOS on Pacific white shrimp juvenile resulted in enhanced the shrimp antioxidative capacity and immunocompetence as well as its robustness against ammonia exposure stress.
“It is even interesting to note that the shrimp survival in treatment MOS12 was not significantly different to that of the negative control and that the immune parameters in the after challenge test were still significantly higher in MOS treatments than those of the control indicating the higher defense capacity of the animals against the disease.”
Along with the immune system stimulating effects, MOS may also play an important role in blocking pathogen colonization in the animal’s GIT.
Torrecillas et al. (2014) suggested that MOS protection against disease outbreaks maybe associated to the promotion of gut pre-epithelial defense mechanism, i.e. by increasing intestinal goblet cells density and mucus production in the intestinal tract.
It has been suggested that mannose could be used as inhibitors of pathogen adhesion to intestinal cells.
“Pathogenic bacteria may recognize and adhere to mannan that mimics the specific carbohydrates groups of intestinal cells. As MOS is not digested, it will be removed with the feces and therefore reducing the incidence of the potential disease.”
In conclusion, the study demonstrated that MOS administration through bioencapsulation in Artemia nauplii at the concentration up to 12 mg MOS L-1 could improve the digestive capacity, growth, survival and immunity of Pacific white shrimp post larvae against V. harveyi infection.
This is a summarized version developed by the editorial team of Aquaculture Magazine based on the review article titled “DIETARY MANNAN OLIGOSACCHARIDES POSITIVELY AFFECT THE GROWTH, DIGESTIVE ENZYME ACTIVITY, IMMUNITY AND RESISTANCE AGAINST VIBRIO HARVEYI OF PACIFIC WHITE SHRIMP (LITOPENAEUS VANNAMEI) LARVAE” developed by: WIDANARNI, ABDURAHMAN TAUFIK, MUNTI YUHANA and JULIE EKASARI – Bogor Agricultural University, Department of Aquaculture, Faculty of Fisheries and Marine Science.
The original article, including tables and figures, was published on ABRIL 2018 through Turkish Journal of Fisheries and Aquatic Sciences.
The full version can be accessed online through this link: http://doi.
org/10.4194/1303-2712-v19_4_01








